The Essential Role of Real World Evidence Combating COVID-19

Are you prepared for potential accelerated approval requirements?
ISPE’s Position on Real World Evidence (RWE)

Read more about the February 2020 position on RWE from ISPE.
Lack of Label Alignment & Patient Safety Implications

Read the full article from UBC’s pharmacovigilance team in Drugs Real World Outcomes.
Risk Management Plan Development & Implementation

Listen to the full podcast from UBC with PharmaVOICE here.
Are You Ready For Sentinel? | New RWE Safety Initiative

With continued investment in the RWE space, find out how to meet the needs of regulators, payers, and prescribers.
Immunogenicity: Meeting the Safety Reporting Requirements
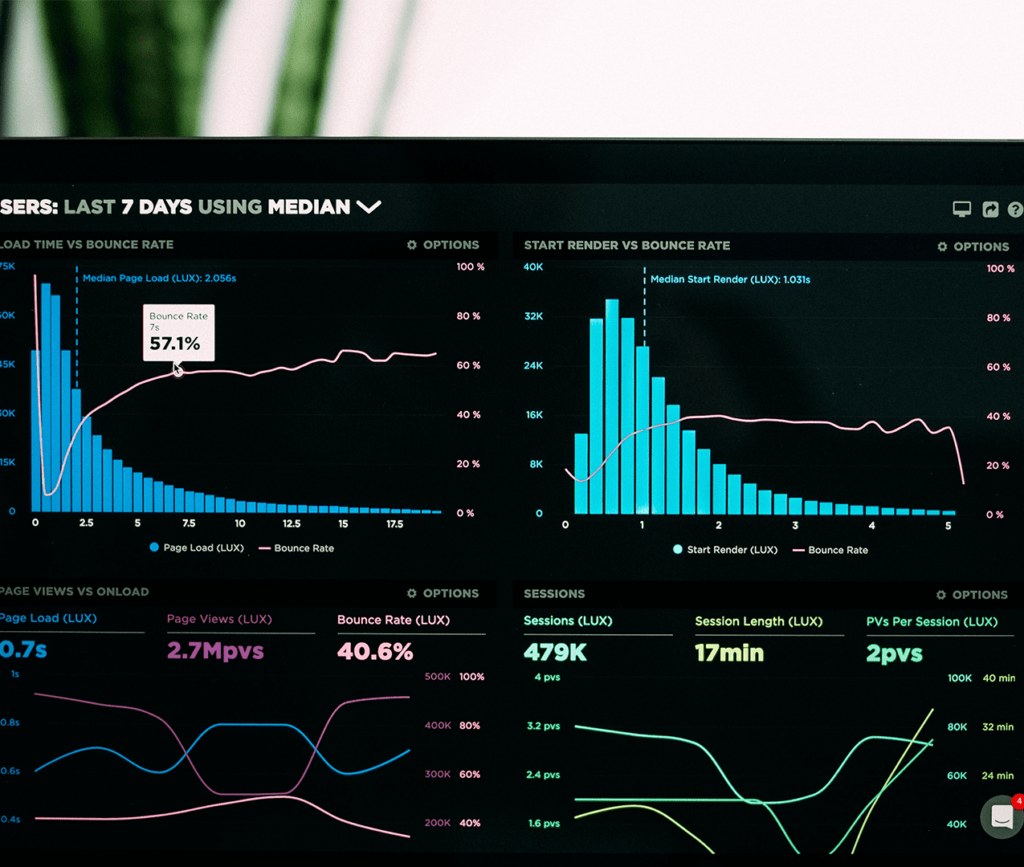
Discover why it is imperative to assess the potential of therapeutic proteins throughout the product lifecycle.
DIA 2019: Watch the Wrap-up of UBC’s Participation

See more of the developments UBC’s evidence development and safety & risk management teams discussed.
Apple’s ResearchKit: The Panacea for Patient Recruitment?

Learn more about this latest offering from Apple and what it could mean for clinical research.




