I. Introduction: The Dual Challenge in Modern Immuno‑Oncology
Immuno‑oncology has reshaped the therapeutic landscape across multiple malignancies, producing durable response in subsets of patients treated with immune checkpoint inhibitors (ICIs).1 These ICIs include anti-programmed cell death – 1 (PD‑1) antibodies nivolumab and pembrolizumab, the anti‑programmed cell death ligand – 1 (PD‑L1) agents atezolizumab and durvalumab, and the anti‑cytotoxic T-lymphocyte-associated protein – 4 (CTLA‑4) antibody ipilimumab.2 Importantly, ICIs function as biomarker‑dependent immunotherapies; their effectiveness is contingent on tumor expression of PD‑1/PD‑L1 or CTLA‑4 pathway activity. Thus, ICIs are administered only to biologically ‘permissive’ tumors that exhibit immune‑evasive signaling. Blocking these inhibitory ligand‑receptor interactions effectively removes tumor‑mediated immune protection and re‑sensitizes malignant cells to T‑cell–mediated killing.3,4 Real‑world effectiveness, however, often falls short of clinical trial efficacy, largely because primary and acquired resistance diminish the durability of response and necessitate earlier transitions to subsequent lines of therapy.5
A potential limitation of some legacy HEOR frameworks could have been their reliance on simplified assumptions of sustained benefit that may not fully capture resistance dynamics. While many contemporary HTA bodies account for waning effects, differences across jurisdictions remain and may lead to value misestimation.
This blog integrates current mechanistic understanding of resistance with its translational consequences for real-world outcomes and discusses pragmatic approaches for embedding resistance biology within HEOR value assessment frameworks.
II. The Biological Architecture of ICI Resistance
Resistance to ICIs reflect a multilayered interplay between tumor-intrinsic features, tumor-extrinsic microenvironmental forces, and host systemic factors.6-8 These mechanisms are not mutually exclusive; they co-evolve, overlap, and may shift over time, driving both primary non-response and secondary escape after initial clinical benefit.
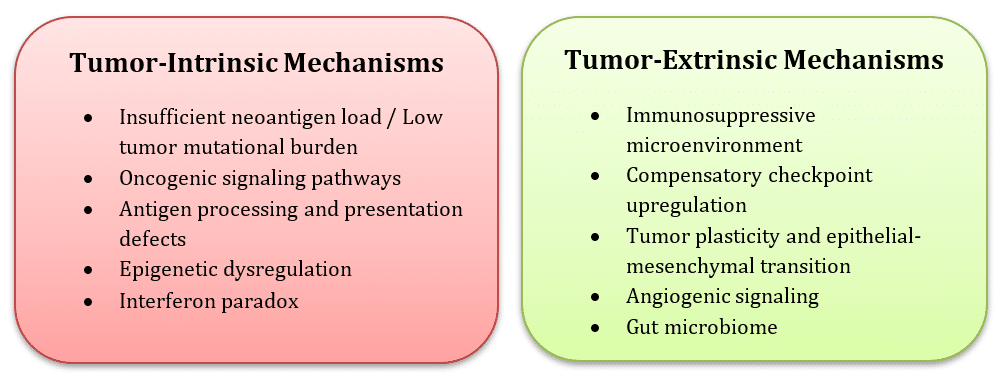
| Mechanisms of ICI resistance: Conceptual illustration created by the authors based on insights from: Pitt et al., 2016; Jenkins et al., 2018; Alsaafeen et al., 2025. |
A. Tumor‑Intrinsic Mechanisms
- Insufficient neoantigen load / low tumor mutational burden (TMB): Diminished immunogenicity impairs antigen-specific priming and cytotoxic T-cell recognition, contributing to primary resistance; clonal evolution can further deplete immunogenic epitopes during therapy.
- Oncogenic signaling pathways: Activation of MAPK, PI3K/AKT, and WNT/β‑catenin pathways promotes immune exclusion, dampens interferon signaling, and increases immunosuppressive signaling, contributing to resistance.
- Antigen processing and presentation defects: Loss of β2-microglobulin and major histocompatibility complex – I (MHC-I) downregulation, or disruption of transporter associated with antigen processing (TAP), abrogate tumor antigen display and blunt ICI efficacy.
- Epigenetic dysregulation: DNA methylation and histone modifications can silence antigen‑presentation pathways and chemokines essential for T‑cell recruitment, contributing to non‑responsiveness.
- Interferon paradox: While acute IFN-γ signaling promotes tumor immunogenicity, chronic signaling can induce epigenomic remodeling, upregulate alternative inhibitory ligands, and drive T-cell exhaustion, culminating in adaptive resistance.
B. Tumor‑Extrinsic Mechanisms
- Immunosuppressive microenvironment: Regulatory T cells (Tregs), myeloid-derived suppressor cells (MDSCs), and tumor-associated macrophages (TAMs) curtail effector T-cell priming and function; impaired STING signaling reduces dendritic cell maturation and T-cell trafficking.
- Compensatory checkpoint upregulation: Post‑PD‑1/PD‑L1 blockade, tumors upregulate TIM‑3, LAG‑3, VISTA, and TIGIT, establishing redundant inhibitory circuits that necessitate combination approaches.
- Tumor plasticity and epithelial–mesenchymal transition (EMT): EMT‑like states confer resistance to cytotoxic T‑cell killing and promote immune exclusion.
- Angiogenic signaling: Vascular Endothelial Growth Factor (VEGF) and hypoxia create endothelial and cytokine barriers to T‑cell infiltration and heighten expression of inhibitory checkpoints on immune cells.
- Gut microbiome: Distinct microbial consortia correlate with response versus resistance; antibiotic exposures and immune toxicities may disrupt microbial ecology and precipitate secondary resistance.
- Host systemic factors: Body composition, diet, physical activity, and sex hormones modulate systemic immunity and may shape immunotherapy outcomes.
The prevalence and timing of resistance mechanisms vary widely by tumor type. Antigen‑presentation defects and interferon‑signaling alterations often emerge early, whereas microenvironmental and checkpoint adaptations typically arise over weeks to months. These uncertainties complicate HEOR translation.7-9
Nevertheless, understanding resistance biology is essential for HEOR because mechanistic heterogeneity directly shapes real‑world durability, survival trajectories, and economic value. These biological insights can help align model structure, biomarker enrichment, and value assessment with the true clinical course, rather than trial‑based idealized assumptions.
III. Translational Consequences: Real‑World Outcomes
Real‑world cohorts consistently demonstrate lower overall survival (OS) and shorter progression‑free survival (PFS) than pivotal clinical trials, reflecting both earlier emergence of resistance during longer real‑world observation periods and the broader clinical characteristics of routine‑practice populations. Differences in observation windows also contribute meaningfully; real‑world cohorts are often followed until death, whereas randomized controlled trials (RCTs) frequently have shorter, protocol‑defined follow‑up periods, limiting detection of late resistance. While real‑world cohorts demonstrate lower population‑level durability due to earlier emergence of resistance and patient heterogeneity, it is important to note that a minority of patients can experience very long‑term benefit. For example, long‑horizon follow‑up of ipilimumab trials in melanoma has shown clear 5‑ to 10‑year survival plateaus, reflecting durable responses that shorter RCT windows may underestimate.10,11 However, these long‑term survivors represent a biologically distinct subgroup and do not diminish the broader resistance‑driven differences observed in real‑world setting.
As resistance emerges, clinicians frequently transition to combination regimens or subsequent therapy lines earlier than anticipated by trial assumptions. Real‑world data in advanced non-small cell lung cancer (NSCLC) (n = 2,631) illustrate this dynamic: outcomes for the full real‑world cohort were markedly inferior to the “trial‑like” subgroup, with shorter OS and PFS, suggesting that resistance‑linked factors such as extensive metastatic burden, suboptimal performance status, or mixed inflammatory signatures accelerate clinical deterioration in broader populations.12 Even meta‑analytic comparisons of RCTs vs. real‑world evidence in melanoma and NSCLC have shown 5–15% lower real‑world response rates, despite similar pooled survival estimates, highlighting how faster clinical decline and non‑standardized monitoring affect real‑world outcomes.13
Consequently, resistance‑driven benefit decay reverberates downstream into survival trajectories, health‑related quality of life, and healthcare utilization. Together, these real‑world patterns underscore the tangible clinical impact of ICI resistance, shaping the durability of benefit and the variability of patient outcomes observed outside controlled trial settings.
IV. Economic Impact of ICI Resistance on Cost‑Effectiveness and Value Assessment
Some economic models for ICIs historically assumed constant hazards, sustained benefit, and fixed post‑progression pathways, yet these assumptions rarely hold in the presence of primary or acquired resistance.14,15 Real‑world decay in treatment effect driven by faster progression, heterogeneous tumor biology, and treatment discontinuation can substantially alter the value proposition of ICIs when compared with trial‑based estimates. More recent cost‑effectiveness studies show that ICIs often fail to meet willingness‑to‑pay (WTP) thresholds when real‑world progression patterns are incorporated. For example, a systematic review of cost‑effectiveness studies across melanoma and lung, head and neck, and genitourinary cancers reported that nivolumab was not cost‑effective in most indications, and pembrolizumab was cost‑effective only in specific biomarker‑selected subgroups, highlighting how resistance‑linked heterogeneity drives large swings in economic value.15 Similarly, economic evaluations in NSCLC reveal that pembrolizumab is cost‑effective only in PD‑L1–high populations, whereas lack of durable response or early resistance substantially worsens cost‑utility outcomes.14 These findings illustrate that while modern HEOR increasingly incorporates real‑world progression patterns, earlier frameworks or non‑mechanistic models may not have fully captured the biological nuances of resistance.
ICI resistance also amplifies incremental cost‑effectiveness ratios (ICERs) by accelerating progression and increasing the need for subsequent therapy, supportive care, and toxicity management. Real‑world durability markedly increases ICERs. For example, perioperative ICI therapy in NSCLC showed ICERs exceeding conventional thresholds in the U.S. and remaining high in middle‑income settings.16 These stem directly from reduced QALY gains when progression occurs earlier than expected due to resistance, alongside high drug acquisition and hospitalization costs.
Further, combination strategies used to overcome resistance are often associated with higher rates of immune-related adverse events, which can lead to long-term morbidity and QALY impacts. Real‑world financial toxicity analyses show that immune‑related adverse events experienced by ~40% of ICI recipients further inflate total care costs and contribute to underestimation of economic burden in traditional models.17
Collectively, these findings suggest that ICI resistance meaningfully diminishes the real‑world clinical and economic value of immunotherapy.
V. Integrating Resistance Biology into HEOR: A Concise Mechanistic Framework
HEOR analysis faces the inherent challenge of translating rapidly evolving biological insights into tractable model structures. The variability and incomplete characterization of resistance mechanisms remain major constraints rather than methodological oversight in HEOR. This blog illustrates these challenges and provides a summary of recommended HEOR approach in context of assessing ICI therapies and resistance development.
Mechanism‑linked real‑world evidence supported by longitudinal biospecimens, molecular profiling, imaging, and toxicity data can more accurately characterize resistance onset, its effect on survival and resource utilization, and the divergence between real‑world and trial‑based durability.18-20 Anchoring model inputs to these biological patterns ensures that value estimates reflect the true clinical course rather than overly optimistic trial assumptions.
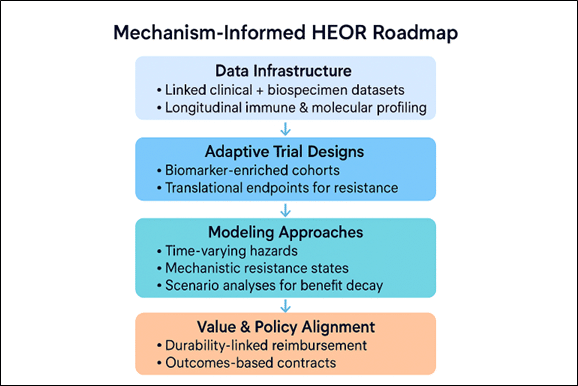
A streamlined operational framework spans four domains: (1) integrated clinical-biospecimen systems capable of tracking longitudinal resistance biology; (2) adaptive, biomarker‑enriched trials evaluating rational combinations and sequencing strategies to overcome resistance; (3) economic models incorporating time‑varying hazards, mechanistic health states, and uncertainty around resistance trajectories; and (4) value and policy mechanisms linking reimbursement to real‑world durability and biomarker‑confirmed benefit.21,22 Collectively, these elements enable HEOR value assessment frameworks to align more closely with the biological realities of ICI resistance and support more sustainable, evidence‑based decision‑making.
VI. Conclusion
Immunotherapy offers transformative benefit, but resistance limits population‑level impact. Integrating tumor, microenvironmental, and host determinants of resistance into evidence generation and economic modeling will improve value assessment and align reimbursement with real‑world effectiveness. As biological insights advance, HEOR frameworks must evolve toward biomarker‑explicit, durability‑focused evaluation.
References
- Kong X, Zhang J, Chen S, et al. Immune checkpoint inhibitors: breakthroughs in cancer treatment. Cancer biology & medicine. 2024;21(6):451-472.
- Park CL, Bedard PL. Moving the needle on immune checkpoint inhibitors with novel targets: are we being TIMid or LAGging behind? The oncologist. 2025;30(7).
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nature Reviews Cancer. 2012;12(4):252-264.
- Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science (New York, NY). 2018;359(6382):1350-1355.
- Berland L, Gabr Z, Chang M, et al. Further knowledge and developments in resistance mechanisms to immune checkpoint inhibitors. Frontiers in immunology. 2024;15:1384121.
- Alsaafeen BH, Ali BR, Elkord E. Resistance mechanisms to immune checkpoint inhibitors: updated insights. Molecular cancer. 2025;24(1):20.
- Pitt Jonathan M, Vétizou M, Daillère R, et al. Resistance Mechanisms to Immune-Checkpoint Blockade in Cancer: Tumor-Intrinsic and -Extrinsic Factors. Immunity. 2016;44(6):1255-1269.
- Jenkins RW, Barbie DA, Flaherty KT. Mechanisms of resistance to immune checkpoint inhibitors. British Journal of Cancer. 2018;118(1):9-16.
- Verdys P, Johansen AZ, Gupta A, et al. Acquired resistance to immunotherapy in solid tumors. Trends in molecular medicine. 2025;31(11):1008-1020.
- Ascierto PA, Del Vecchio M, Mackiewicz A, et al. Overall survival at 5 years of follow-up in a phase III trial comparing ipilimumab 10 mg/kg with 3 mg/kg in patients with advanced melanoma. Journal for immunotherapy of cancer. 2020;8(1):e000391.
- Giacco V. Long-term results with nivolumab and ipilimumab in melanoma. Nature Cancer. 2025;6(3):407-407.
- Ge W, Wu N, Jalbert JJ, et al. Real-World Outcomes and Prognostic Factors Among Patients with Advanced Non-Small Cell Lung Cancer and High PD-L1 Expression Treated with Immune Checkpoint Inhibitors as First-Line Therapy. Cancer management and research. 2022;14:3191-3202.
- Digkas E, Tabiim AJ, Smith D, Valachis A. Randomized Versus Real-World Evidence on the Efficacy and Toxicity of Checkpoint Inhibitors in Cancer in Patients with Advanced Non-small Cell Lung Cancer or Melanoma: A Meta-analysis. Targeted oncology. 2022;17(5):507-515.
- Ding H, Xin W, Tong Y, et al. Cost effectiveness of immune checkpoint inhibitors for treatment of non-small cell lung cancer: A systematic review. PloS one. 2020;15(9):e0238536.
- Verma V, Sprave T, Haque W, et al. A systematic review of the cost and cost-effectiveness studies of immune checkpoint inhibitors. Journal for immunotherapy of cancer. 2018;6(1):128.
- Aranishi T, Fujioka M, Shukuya T, et al. 611P Cost-effectiveness of perioperative ICI therapy with NSCLC in Japan, USA, Brazil. Annals of Oncology. 2024;35:S1621-S1622.
- Liu J, Lustberg M, Sonis S. Improving assessment of financial toxicity of cancer immunotherapy. Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer. 2025;33(8):735.
- Güven DC, Thong MS, Arndt V. Survivorship outcomes in patients treated with immune checkpoint inhibitors: a scoping review. Journal of cancer survivorship : research and practice. 2025;19(3):806-845.
- Liu SJ, Yan LJ, Wang HC, et al. Safety, efficacy, and survival outcomes of immune checkpoint inhibitors rechallenge in patients with cancer: a systematic review and meta-analysis. The oncologist. 2024;29(11):e1425-e1434.
- Nieder C, Aanes SG, Stanisavljevic L, Mannsåker B, Haukland EC. Real-World Survival Outcomes in Patients with Different Types of Cancer Managed with Immune Checkpoint Inhibitors. Oncology research and treatment. 2025;48(3):82-91.
- Coschi CH, Juergens RA. Overcoming Resistance Mechanisms to Immune Checkpoint Inhibitors: Leveraging the Anti-Tumor Immune Response. Current oncology (Toronto, Ont). 2023;31(1):1-23.
- Santiago-Sánchez GS, Fabian KP, Hodge JW. A landscape of checkpoint blockade resistance in cancer: underlying mechanisms and current strategies to overcome resistance. Cancer biology & therapy. 2024;25(1):2308097.
About UBC
United BioSource LLC (UBC) is the leading provider of evidence development solutions with expertise in uniting evidence and access. UBC helps biopharma mitigate risk, address product hurdles, and demonstrate safety, efficacy, and value under real-world conditions. UBC leads the market in providing integrated, comprehensive clinical, safety, and commercialization services and is uniquely positioned to seamlessly integrate best-in-class services throughout the lifecycle of a product.


About the Author
Gaurang Nazar, MD, PhD, Senior Associate Research Scientist
With extensive research experience, Gaurang has focused on non-communicable disease (NCD) prevention and control, as well as the Health Economics and Outcomes Research domain. His expertise encompasses research design, methods, data analysis, and scientific writing. He has actively contributed to numerous research projects and publications, consistently delivering high-quality outcomes. Gaurang has served as an investigator on NCD research projects funded by esteemed agencies such as the National Institutes of Health (NIH), Wellcome Trust, UK Medical Research Council (MRC), UK Research and Innovation (UKRI), and the World Health Organization (WHO). Gaurang’s contributions to Evidinno encompass a range of medical disciplines, and his involvement in prestigious research projects highlights his dedication to advancing scientific knowledge and improving health outcomes. Gaurang holds a PhD in Epidemiology from the London School of Hygiene and Tropical Medicine, University of London (UK), a Master of Science (Msc) in Population Health from the University of Wollongong, Australia, and a Bachelor of Medicine and Bachelor of Surgery (MBBS) in Clinical Medicine from South Gujarat University, India.





